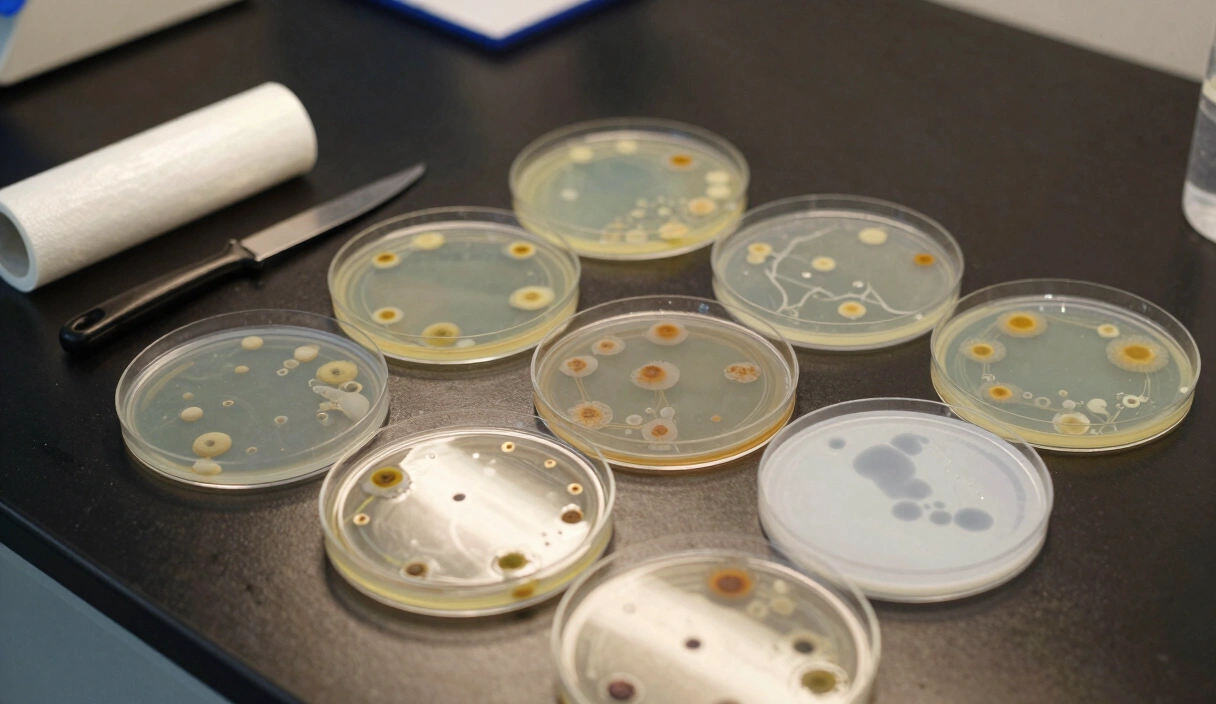
MEA vs. PDA: The Professional Laboratory Guide to Mushroom Agar Recipes
MEA, PDA, and antibiotic agar — five tested recipes with exact measurements for isolating clean mushroom cultures on plates.
Contents
Around 600 AD, cultivators in Ancient China figured out that Wood Ear mushrooms would reliably colonize freshly cut logs if they placed the logs near existing fruiting bodies. No agar, no sterile technique, no pressure cookers. Just proximity and patience. Fourteen centuries later, agar recipes for mushrooms have replaced those rotting logs with petri dishes and precisely calibrated nutrient gels, but the fundamental principle hasn’t changed: give the fungus what it wants, and it will grow.
The difference now is precision. Choosing a recipe is not “mixing powder and water.” You are balancing the Carbon-to-Nitrogen ratio of the medium, managing the Maillard Reaction during sterilization, and steering mycelial morphology toward aggressive rhizomorphic growth. Get the chemistry wrong, and your plates turn brown, your genetics stall, and you’ve wasted an afternoon inside a Still Air Box wondering where it all went sideways. This guide covers the molecular mechanisms behind MEA, PDA, and selective antibiotic media, with the protocols I actually use in my own lab.
The Chemistry of the Medium: Carbohydrates vs. Nitrogen
A fungal agar medium consists of three primary components: the structural matrix (Agar), the energy source (Carbohydrates), and the building blocks (Nitrogen).
1. The Agar Matrix
Agar-agar is a gelatinous polysaccharide derived from red algae. It provides zero nutritional value to the fungus; its sole purpose is to provide a semi-solid surface that prevents bacterial motile expansion while allowing fungal hyphae to colonize in two dimensions.
2. The Carbon Source (LME vs. Dextrose)
- Light Malt Extract (LME): Contains a complex mix of maltose, glucose, and dextrins. It encourages strong, exploratory Rhizomorphic growth.
- Dextrose: A simple sugar that provides a rapid energy spike. It is the primary fuel in PDA (Potato Dextrose Agar).
3. The Nitrogen Factor (Peptone & Yeast Extract)
Standard LME agar is nitrogen-poor. To accelerate growth, professionals add Peptone (hydrolyzed protein) or Yeast Extract.
- The Rationale: Nitrogen is the limiting factor for protein synthesis. By adding 1g of Peptone per liter, you can reduce colonization time by up to 30%, as the fungus doesn’t have to synthesize its own amino acids from scratch.
The Sterilization Rule: Avoiding the Maillard Reaction
A common point of failure in home labs is over-sterilizing the media. When carbohydrates (sugars) and nitrogen sources (amino acids) are heated together under pressure, they undergo the Maillard Reaction.
I ruined 40 plates in a single batch last February because I left the Presto 23-quart at pressure for 45 minutes instead of 20. Every plate came out the color of dark maple syrup.
Why It Kills Your Mycelium
The Maillard reaction creates brown nitrogenous polymers called melanoidins. Not only does this reduce the nutritional value of your sugar, but at high concentrations, these byproducts can become inhibitory or toxic to sensitive mycelium.
- The Technical Protocol: Sterilize your agar at 15 PSI (121°C) for exactly 20 minutes.
- The Cool Down: Remove the flask as soon as the pressure hits zero. Leaving the media in a hot pressure cooker for an extra hour will lead to “caramelization,” turning your clear agar a dark, muddy brown and stunting your genetics.
Professional Agar & Lab Tools

Mushroom Agar According to Kimmig (Pack of 20)
Pre-poured sterile agar plates optimized for fungal mycelium growth.
Check Price on Amazon
Sabouraud 2% Glucose Agar Plates (Pack of 20)
Sterile nutrient media plates for advanced microbiological cultures.
Check Price on Amazon
Nutrient Agar Petri Dishes Science Kit
Complete agar kit for home mycology experiments and culture isolation.
Check Price on Amazon* Affiliate links. Prices last updated March 6, 2026.
Top 3 Industry-Standard Agar Recipes (per 500ml)
For precision work, always mix your media using distilled water and a digital scale accurate to 0.01g.
1. MEA (Malt Extract Agar) – The General Specialist
The most versatile recipe for gourmet Basidiomycetes.
- 500ml Distilled Water
- 10g Agar-Agar Powder
- 10g Light Malt Extract (LME)
- 1g Peptone (Optional, for growth boost)
2. PDA (Potato Dextrose Agar) – The Vigor Booster
Excellent for “lazy” or weak cultures that need high-calorie fuel.
- 500ml Potato Infusion (The water from boiling 150g of sliced potatoes for 20 mins)
- 10g Agar-Agar Powder
- 10g Dextrose
I used to think PDA was strictly inferior to MEA. Then I ran side-by-side transfers of a sluggish Lion’s Mane isolate and the PDA plates outpaced the MEA plates by 3mm/day in radial expansion. Species matters more than recipe loyalty.
3. Charcoal Agar – The Diagnostic Master
By adding 1g of Activated Charcoal to the MEA recipe, the medium turns pitch black.
- The Advantage: Mycelium is white; mold is often grey or green. Contamination becomes visible on a black background days earlier than on clear agar. This is the gold standard for “cleaning” wild tissue clones.
Selective Media: The Antibiotic Protocol
When cloning mushrooms from the wild or working with “dirty” spore prints, bacteria are your primary competitor. To eliminate them, we use Selective Media.
The Gentamicin Standard
Gentamicin Sulfate is the preferred antibiotic because it is heat-stable—it can be added to the flask before autoclaving.
- Dosage: 0.04g per Liter (40mg/L).
- The Effect: This concentration inhibits a wide spectrum of Gram-positive and Gram-negative bacteria without hindering the growth of the fungal dikaryon.
I watched a grower on a forum dump 400mg/L of gentamicin into his plates because he figured “more antibiotic equals more protection.” He killed every culture he put on them. That $85 worth of Sigma-Aldrich gentamicin went straight into the trash.
Pouring Technique: The 47°C “Sweet Spot”
The final technical hurdle is Condensation. If you pour your agar while it is boiling hot, the lids will fog up, creating water droplets that can harbor bacteria.
- The Rule: Wait until the flask reaches 117°F (47°C). At this temperature, the agar is still liquid but cool enough that it won’t produce excessive steam.
- The Stack Method: Stack your petri dishes 5–10 high and pour from the bottom up. The warmth from the dish above prevents the lid of the dish below from fogging.
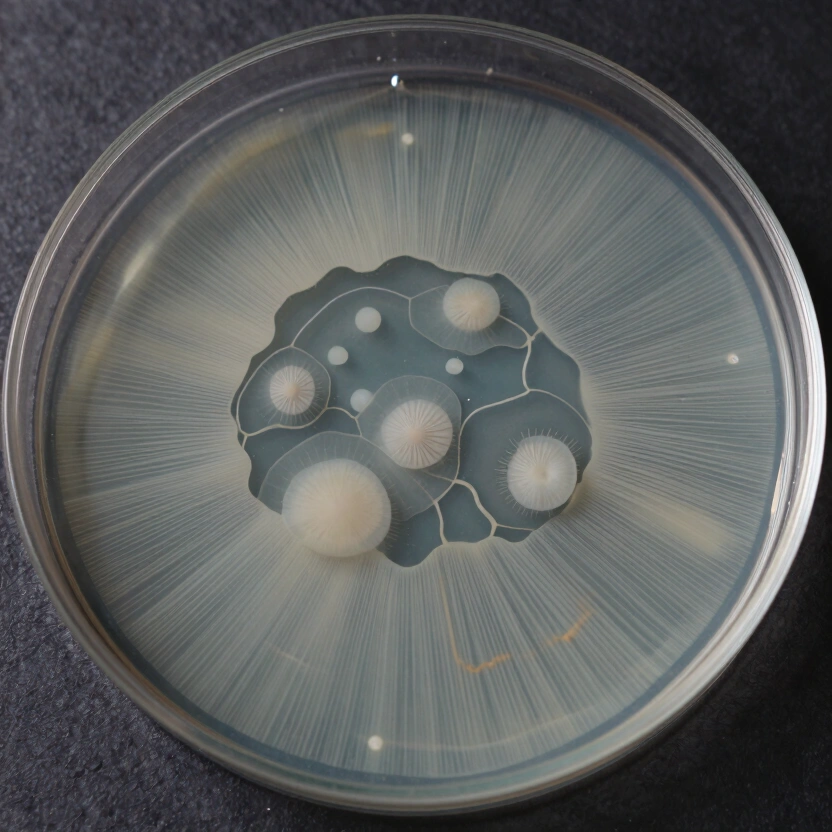
If your agar keeps caramelizing after 20 minutes at 15 PSI, check your pressure cooker’s gauge calibration before blaming the recipe. Once your plates are clean and poured, the next step is isolating high-performance sectors on those plates to lock in the genetics that actually produce.
Frequently Asked Questions
Why won’t my agar solidify after autoclaving?
Low pH is the usual culprit. Acidic tap water or fruit-juice additives prevent the polysaccharide chains from bonding. Over-sterilization does the same thing: cooking agar for 60+ minutes breaks those chains through acid hydrolysis, leaving you with a liquid syrup that never sets.
Can I substitute gelatin for agar in mushroom plates?
Gelatin is animal collagen, and most bacteria and fungi produce proteases that digest it within days. Your solid plate turns to liquid. Gelatin also melts at 77 degrees F, which is standard incubation temperature, so the plate would collapse even without enzymatic attack. Agar stays solid up to 185 degrees F and is indigestible by most fungi.
How long can I store poured agar plates before they go bad?
Wrap them in Parafilm, seal them in a Ziploc, and refrigerate. They last 3 to 6 months.
What does sectoring on an agar plate mean?
A dikaryon often shows different growth patterns radiating outward: one sector fuzzy and slow (tomentose), another fast and rope-like (rhizomorphic). By taking a small biopsy from the most rhizomorphic sector and transferring it to a fresh plate, you select for speed and yield. This is the foundation of strain isolation, and understanding it will change how you manage your genetic library.
Does food coloring in agar affect mycelium growth?
It does not. Blue for MEA, red for PDA is a common organizational convention that also makes the fine, translucent leading edge of mycelium easier to spot during transfers.
Related Articles
Mushroom Agar Work: The Complete Guide to Isolating, Storing, and Expanding Cultures
Master the "black belt" of mycology. From Fanny Hesse’s kitchen discovery to advanced genetic sectoring, we explore the physics, chemistry, and real-world hacks of agar work.

Top Mushroom Cultivation Techniques: A Technical Efficiency Comparison
Monotub, Bucket Tek, or PF Tek? A side-by-side comparison using Biological Efficiency and yield per square foot to find the best method for your space.

The Ultimate Mushroom Substrate Guide: Engineering High-Yield Growth Media
Not all substrates work for all species. C:N ratios, pH buffering, moisture content, and specific recipes matched to the mushrooms you're actually growing.