Strain Isolation and Sectoring: Mastering Mycelial Purity and Longevity
How to isolate fast-growing mushroom genetics on agar through sectoring. Includes transfer protocols and signs of mycelial senescence.
Contents
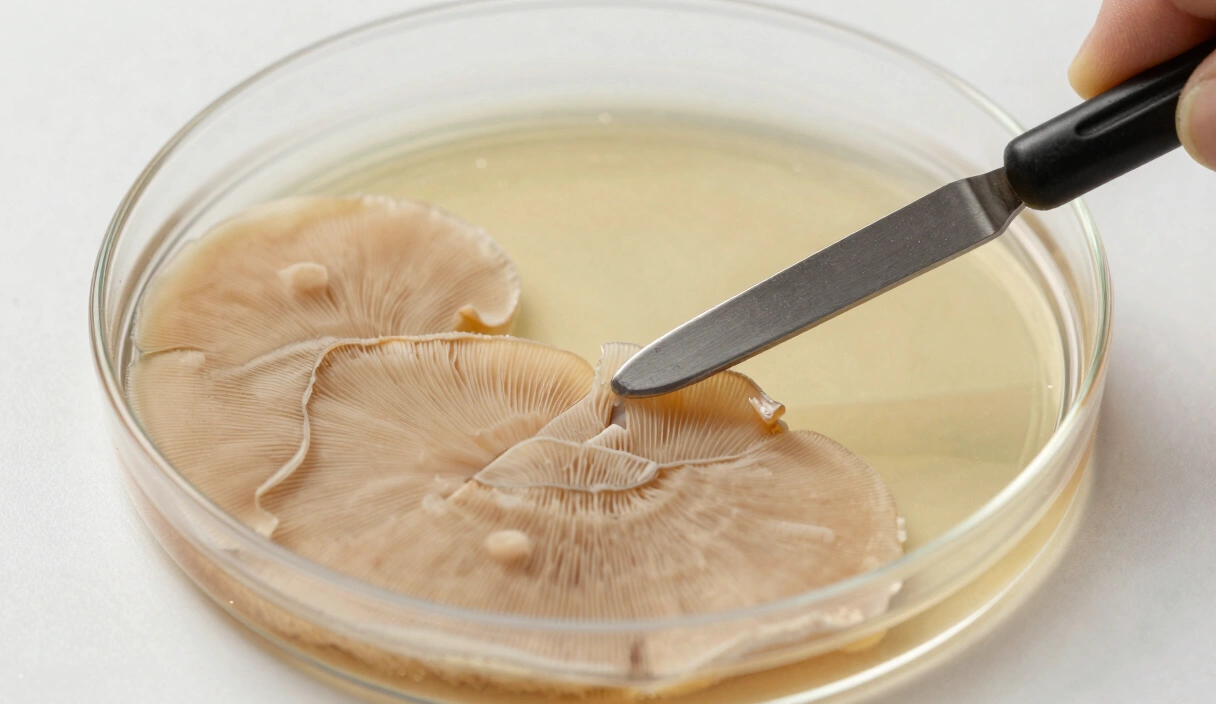
The scalpel tip hovers a millimeter above the agar surface. Under the SAB, my arms are cramping, and the isopropyl fumes are making my eyes water. But this transfer matters. The rhizomorphic sector on plate G3-07 has been outpacing everything else by 4mm/day, and I need a clean biopsy from its leading edge before the tomentose growth behind it catches up and muddies the genetics. This is strain isolation and sectoring in practice: not a textbook exercise, but a timed operation where sterile technique and pattern recognition collide.
Strain isolation is the technical art of identifying and isolating the most aggressive, productive segments of a fungal colony on agar. It turns a multispore “genetic lottery” into a repeatable, commercial-grade culture. But this is not a process of infinite expansion. You are fighting Mycelial Senescence, a biological expiration date driven by mitochondrial decay and oxidative stress. To reach 100% Biological Efficiency consistently, you must learn to read the mycelium’s growth patterns on the plate, identify high-performance sectors, and implement a genetic management protocol that prevents your strains from aging into obsolescence.
The Biology of Selection: Fighting Senescence
A common misconception in mycology is that mycelium is immortal. It is not. Fungi, particularly Basidiomycetes, are subject to a phenomenon analogous to the Hayflick Limit in human cells: Senescence.
The Mechanism of Decay
As mycelium expands, its cells undergo constant division. Each division introduces a risk of mitochondrial DNA damage. Over time, this damage accumulates, leading to:
- Reduced Colonization Speed: The “Leap-off” time increases from 48 hours to 7+ days.
- Enzymatic Exhaustion: The fungus loses its ability to effectively degrade lignin and cellulose.
- Sporulation Failure: The mycelium may colonize the block but refuse to produce primordia.
The Golden Rule of Transfers: To maintain genetic vigor, never perform more than 8 to 10 serial transfers from an active plate before returning to your genetic “Master” (a culture stored in a cold-dormancy state).
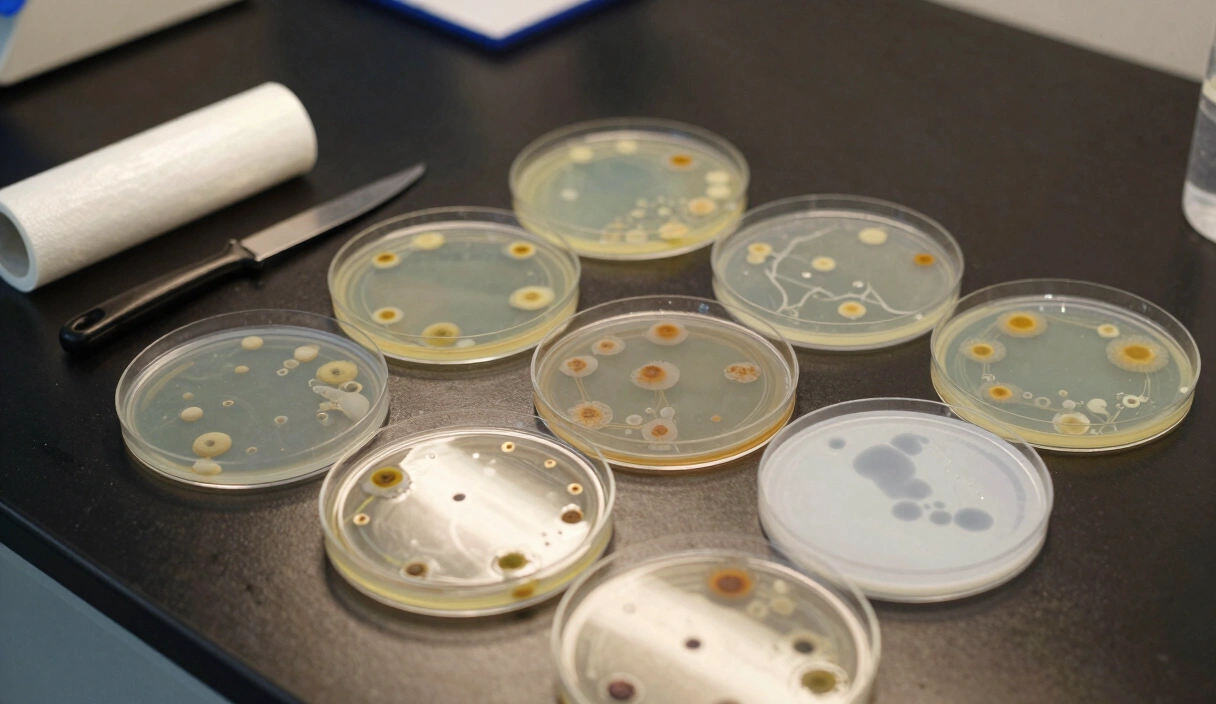
Sectoring: Reading the Mycelial Map
When you germinate spores on an agar plate, you aren’t growing one organism; you are growing a chaotic battlefield of thousands of different “mating pairs,” each with its own genetic potential.
In my tracking spreadsheet from 2024, out of 36 multispore plates I poured for Blue Oyster, only 4 produced a sector with colonization rates above 6mm/day. That’s an 11% hit rate. The other 32 plates produced acceptable but unremarkable genetics.
Identifying a Sector
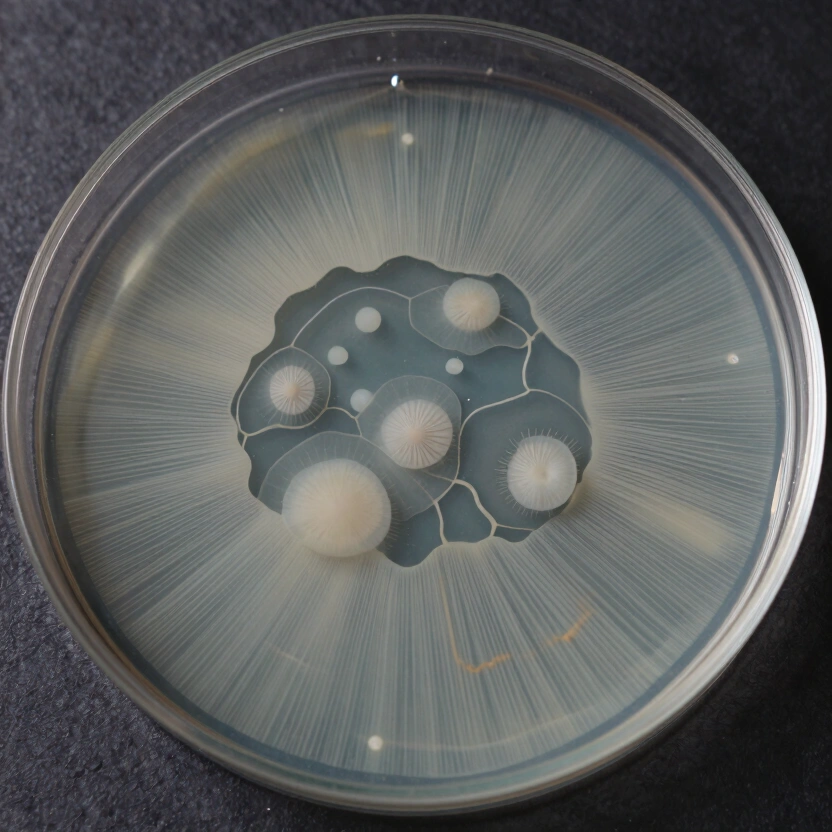
Look for distinct visual boundaries on your agar plate where one growth pattern meets another. This is a Sector.
- The Rhizomorphic Sector (The Goal): Appears as thick, rope-like strands that radiate outward like miniature lightning bolts. This morphology is organized for high-speed nutrient transport and is the primary marker of high-yield genetics.
- The Tomentose Sector (The Backup): Appears as soft, cotton-like fuzz. While not necessarily low-yield, tomentose growth is often slower and can mask subtle bacterial contamination.
- The “Slow” Sector: A flat, translucent zone that lags behind the rest of the plate. This indicates weak genetics or high metabolic stress. Never transfer from these zones.
Advanced Laboratory Isolation Tools

Mushroom Agar According to Kimmig (Pack of 20)
Pre-poured sterile agar plates optimized for fungal mycelium growth.
Check Price on Amazon
Sabouraud 2% Glucose Agar Plates (Pack of 20)
Sterile nutrient media plates for advanced microbiological cultures.
Check Price on Amazon
Nutrient Agar Petri Dishes Science Kit
Complete agar kit for home mycology experiments and culture isolation.
Check Price on Amazon* Affiliate links. Prices last updated March 6, 2026.
The Technical Workflow: From Spores to Isolated Dikaryon
Phase 1: The Spore Streak
Using a sterile loop in your Still Air Box (SAB), streak a minute amount of spores across an MEA agar plate. Wait 5–7 days for germination. You will see a “messy” plate with hundreds of tiny white specks.
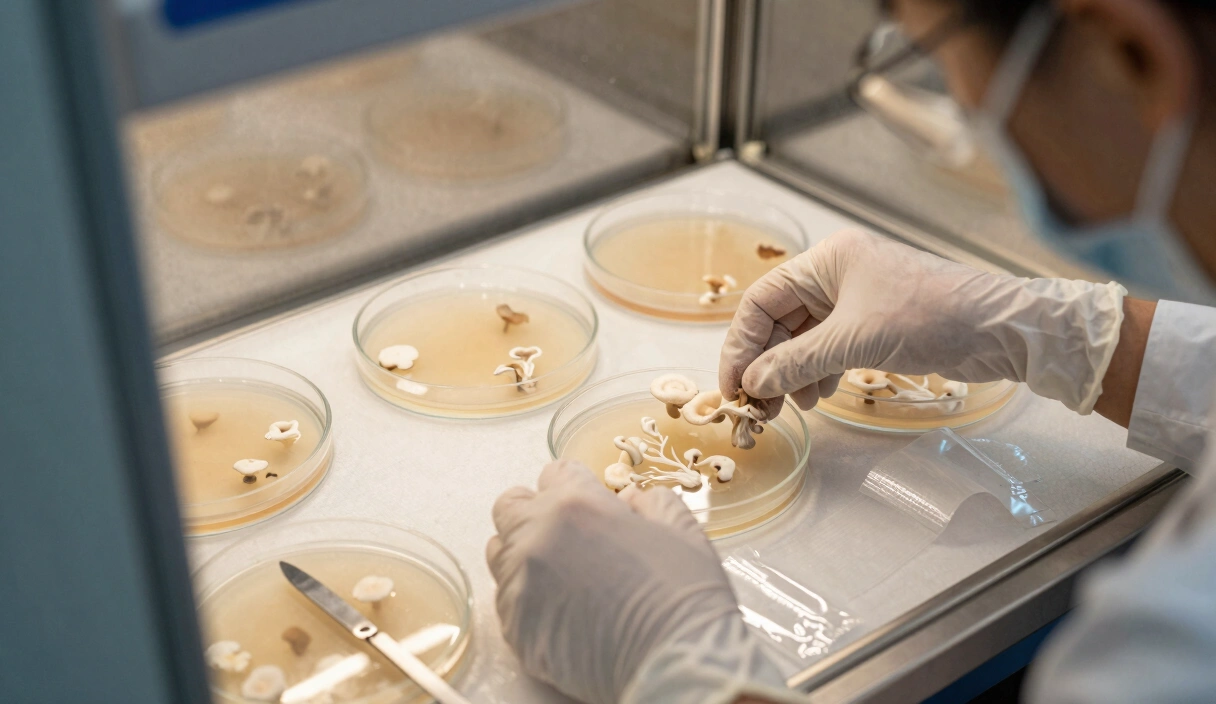
Phase 2: The First Cleanup
Identify the three or four areas of the plate that germinated first. These are your fastest “early-start” genetics. Use a scalpel to take a 2mm x 2mm biopsy from the leading edge of these colonies and move them to fresh plates.
A 2019 study in Fungal Genetics and Biology measured a 47% decline in colonization speed after just 15 serial transfers in Pleurotus ostreatus. Fifteen. If you are not tracking your transfer count, you are flying blind toward senescence.
Phase 3: Isolating the Rhizomorphs
On your second-generation plates, you will begin to see Sectoring. One side of the biopsy may grow fuzzy, while the other grows rope-like.
- The Precision Transfer: Take a biopsy only from the most aggressive, rhizomorphic tip.
- Consistency Check: By the third or fourth transfer, the entire plate should display a uniform, circular, rhizomorphic growth pattern. You have now isolated a Dikaryotic Strain.
Genetic Architecture: Master Slants vs. Working Plates
If you continue to transfer from your working plates indefinitely, your strain will undergo Genetic Drift and senescence. To prevent this, you must build an archive.
The Master Slant Protocol
A Master Slant is a test tube containing a specific “high-grip” agar recipe (often supplemented with wood or grain) where the mycelium is allowed to grow and then placed into a state of Cold Dormancy (38°F / 4°C).
- The Workflow: Create 10 slants from your first successful isolation. Use one slant to create your “working plates.” When that slant is exhausted or the working strain shows signs of slowing down, open the second master slant.
- Longevity: Correctly prepared master slants can preserve a high-yield strain for 2 to 5 years with zero genetic degradation.
Most guides tell you to make 3 master slants per strain. That number is too low. Commercial labs at Fungi Perfecti maintain 10 to 12 slants per production strain because the failure rate on revival after 2+ years of cold storage hovers around 15-20%.
Senescence Diagnosis Table
| Symptom | Diagnosis | Recommended Action |
|---|---|---|
| Translucent Leading Edge | Early Senescence | Return to Master Slant or re-isolate from spores. |
| Brown Metabolites on Agar | High Metabolic Stress | Adjust incubation temp; check for bacterial hitchhikers. |
| “Aerial” tomemtose growth | Lost Directionality | Re-isolate rhizomorphic sectors from the leading edge. |
| No “Leap-off” on Grain | Terminal Senescence | Discard culture; start over from a clone or spore bank. |
Once your third-generation plate shows uniform radial symmetry, the genetics are locked in, but they won’t stay that way without proper storage. Build your Master Slant archive before the senescence clock runs out, and when you are ready to scale beyond agar, liquid culture will turn a single plate into enough inoculant for 50 grain jars.
Frequently Asked Questions
Will switching agar recipes fix a strain that’s slowing down?
Temporarily, at best. Moving a tired culture from MEA to PDA can produce a short-term vigor boost through a “Media Shift” effect, but it does not repair accumulated mitochondrial damage. The culture usually stalls again during the bulk fruiting phase. Return to a dormant master culture or restart from spores for a permanent fix.
How can I tell if my agar isolation is actually pure?
Look for radial symmetry. A pure dikaryotic isolation grows outward from the center in an even circle. Lopsided growth or uneven thickness means competing strains are still present and you need at least one more transfer.
Is rhizomorphic mycelium always better than tomentose for yields?
About 90% of the time. Rhizomorphic growth moves nutrients over distance more efficiently, which is why it colonizes bulk substrates faster. But some species are naturally tomentose, including certain Reishi varieties, so research your specific species before discarding a fuzzy isolate. Check Paul Stamets’ species-specific notes in Mycelium Running if you are unsure.
How many genetic backups should I keep of each strain?
Follow the Rule of Three: one Master Slant in your primary fridge, one backup slant in a separate location, and one culture stored in sterile distilled water, which can last 10+ years for some species.
Should I start isolation from a clone or from spores?
Spore isolation gives you high genetic diversity but requires multiple transfers to find a strong strain. Cloning captures proven genetics instantly from a large, healthy mushroom. The tradeoff: clones start further along the senescence clock, so they demand stricter transfer management from day one.
Related Articles
Mushroom Agar Work: The Complete Guide to Isolating, Storing, and Expanding Cultures
Master the "black belt" of mycology. From Fanny Hesse’s kitchen discovery to advanced genetic sectoring, we explore the physics, chemistry, and real-world hacks of agar work.
MEA vs. PDA: The Professional Laboratory Guide to Mushroom Agar Recipes
MEA, PDA, and antibiotic agar — five tested recipes with exact measurements for isolating clean mushroom cultures on plates.

The Monotub Masterclass: Engineering Fresh Air Exchange for High-Yield Grows
Everything about monotub design — hole placement, polyfill density, and the convection physics that make or break your first bulk grow.